Building Medical Device Innovations Together
Your Objective,
Your Success
Graphics Designed by macrovector / Freepik
We specialize in partnering with medical device and biotechnology companies through a tailored approach that helps these companies understand and achieve their ultimate business goals in regulatory, quality, and reimbursement strategies.
What Are Your Objectives?
We are more than just consultants, medical writers, or regulatory specialists working on a project for you. Instead, we partner with you to ensure our services are customized to your needs. Think of us as an extension of your team, helping you every step of the way to achieve your own telos– or ultimate goal.
Regulatory & Quality Solutions
We demystify the complexities of regulatory pathways and quality systems to provide a concrete path-forward.
Clinical Adoption
Utilizing a full range of scientific tools, we create and communicate evidence to demonstrate your product’s value proposition.
Reimbursement Strategies
Our strategic approach provides clear steps to reimbursement by defining the medical needs, clinical benefits, and economic insights for payers.
Trusting in Telos is Telos trusting in your business – because when we help your business advance, we help improve the quality of patient care.
Telos by the Numbers
Years of Regulatory & Quality Experience
Years of Cumulative Experience Collaborating with Clinicians/KOLs on Pre-Clinical and Clinical Science
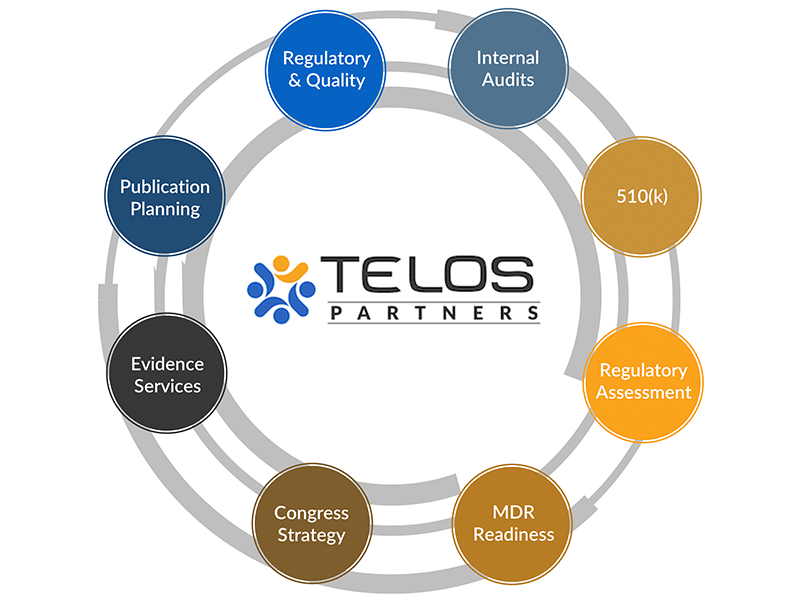
Our Services Capabilities Integrate to Provide the Best Possible Solution and Service Combination for our Clients.
“Telos pulled us out of the fire a bunch of times. There were times that I called Telos and said that I need this and this by this date… and they would always deliver. Telos’ flexibility and their ability to hit deadlines to turn things around quickly was a game changer.”
Four Steps to the
“Ultimate Objective”
As your partner, Telos invests time and effort from the onset of connection to understand your ultimate business objectives. This approach helps us to prescribe and provide the right services for success. Telos means “ultimate objective” – let us help you reach yours.
Define Your Goals
Telos works with you to clearly understand your business’ goals. We ask “Why” instead of “How.”
Identify Services & Plan
Our team identifies service areas where we can partner to create a plan unique to your business.
Deliver Solutions
We’ll work with you through each phase to accomplish the milestones identified in the plan.
Repeat as Business Scales
Because we see each client as a true partner, we remain ready to help your business scale as new needs arise.
How We Meet Your Objectives
EVIDENCE SERVICES
Your products’ success starts with the foundations of pre-clinical, clinical, and health economics evidence.
We leverage a wide range of analytical approaches and forums of communication to convert your data to disseminated evidence.
REGULATORY & QUALITY SOLUTIONS
Whether seeking novel indications or aiming for first-time marketing approval, our expert regulatory and quality teams will help you win in a competitive landscape.
Our experts provide your team access to over 40 years of regulatory and quality subject matter expertise globally and ensure you have round-the-clock compliance to stay ahead.


Why An Integrated Approach?
Our evidence, regulatory, and quality services operate in a unique, integrated fashion to achieve your ultimate goals by circling-up around each objective.
Instead of providing services, we work to provide intrinsic value to your success. View our case studies to see how we’ve helped clients just like you:
Start the Conversation
Our Clients and Partners