Building Medical Device Innovations Together
Our clinical research services support the conversion of data to evidence and sharing that evidence broadly to communicate the value proposition of your technology.
No data you say? Our contract research organization (CRO) provides a full suite of clinical research services to conduct business-balanced studies that are designed and executed to meet the specific needs.
Clinical Research Services
Regardless of your clinical research needs, our contract research organization (CRO) experts take a strategic approach to study design and conduct that balances the needs with appropriate investment. Our research professionals bring unique solutions, derived from years of experience and out-of-the-box thinking, to provide tailored services, such as:
- Study design and regulatory (e.g. IDE, IND)
- Clinical databases
- Monitoring, data capture, and reporting
- Retrospective or prospective studies
- Domestic and/or outside U.S. studies
- Single-arm, registry, or randomized studies
- Biostatistics and adaptive designs
- Post-market clinical follow-up studies
Telos strives to be more than just “a contract research organization (CRO).” We put ourselves in your shoes to ensure our services are in lockstep with your goals as if Telos is an extension of your business and a part of your team.
Our Unique Approach Combined with Years of Expertise Provides Our Clients With Solutions They Can Trust and Scale
Telos by the Numbers
Years of Cumulative Experience Collaborating with Clinicians/KOLs on Pre-Clinical and Clinical Science
Our Clinical Research Services Integrate with Many Telos Solutions to Provide the Best Possible Solutions and Service for Our Clients
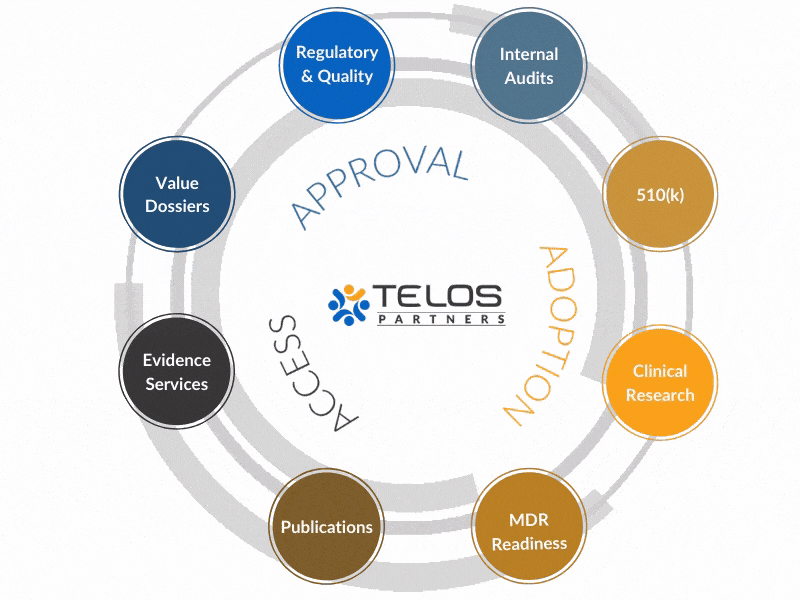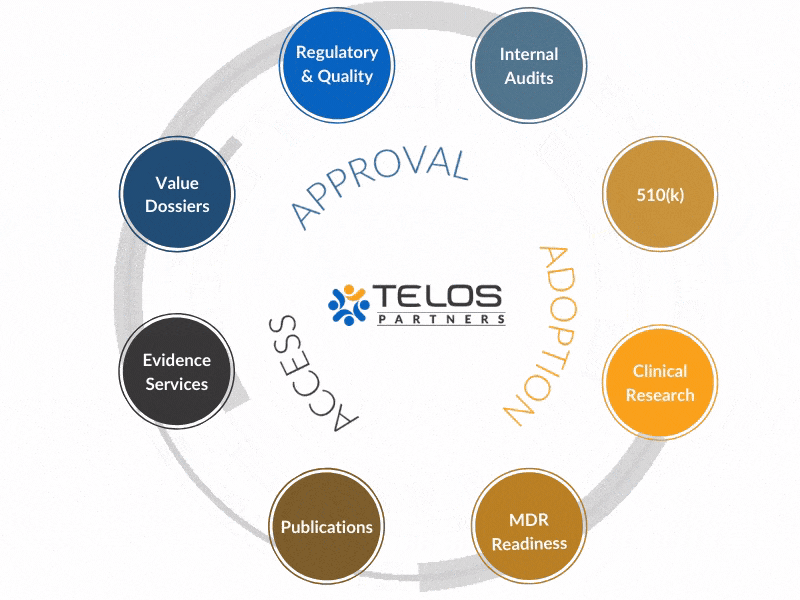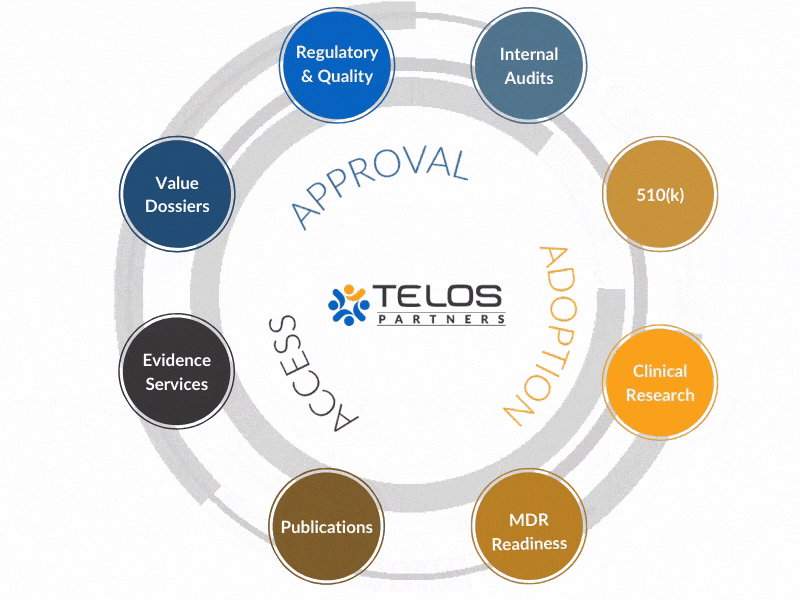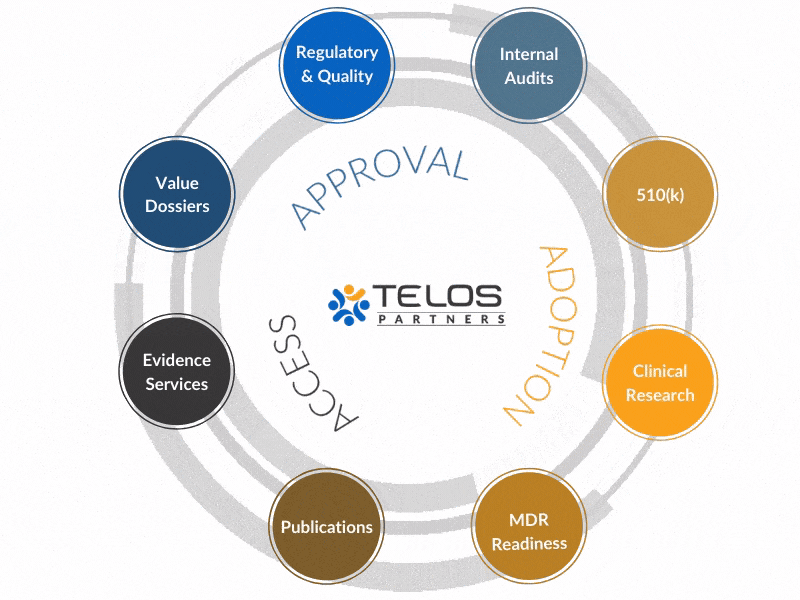
Case Study:
“We need to gather clinical data through a retrospective study to meet the needs of publication for market access and clinician adoption. We also need our initial CE Mark.”
Client: Spinal implant devices
Solution Areas Identified: Clinical research, regulatory and quality services, evidence services.
Objectives Met: On-site monitoring and CRO support for each clinical site, CE Mark approval in process, and plans for additional prospective registry study to generate additional post-market clinical follow-up evidence.
Bonus Objectives Identified and Achieved: The results of the clinical study were published in the International Journal of Spine Surgery thanks to the collaboration with the surgeon investigators. This published evidence will foster further adoption in the United States and support market access outside of the United States.
Four Steps to the
“Ultimate Objective”
As your partner, Telos invests time and effort from the onset of connection to understand your ultimate business objectives. This approach helps us to prescribe and provide the right services for success. Telos means “ultimate objective” – let us help you reach yours.
Define Your Goals
Telos works with you to clearly understand your business’ goals. We ask “Why” instead of “How.”
Identify Services & Plan
Our team identifies service areas where we can partner to create a plan unique to your business.
Deliver Solutions
We’ll work with you through each phase to accomplish the milestones identified in the plan.
Repeat as Business Scales
Because we see each client as a true partner, we remain ready to help your business scale as new needs arise.

